Two components of electrical dark noise in toad retinal rod outer segments. 1225) confirm that isomerization is the rate-limiting step controlling thermal noise, and they provide a molecular understanding of the Barlow correlation. Thermal activation of the visual transduction mechanism in retinal rods. Membrane current wasrecorded from intact, isolated rods ortruncated, internally dialyzed rod outer segments. We investigated the molecular mechanism of the continuous component of the electrical dark noise in toad rods. A second cordinate BLA (corresponding to the elongation of double bonds coupled with a shortening of single bonds and connecting TSCT, CI, and TSDIR) is shown in addition to RC. A recent study suggested that thermal activation involves a canonical isomerization reaction. ABSTRACT Noise in the rod photoreceptors limits the ability of the dark-adapted visual system to detect dim lights. (A) Schematic overview of the S0 potential energy surface driving the thermal isomerization of bovine Rh, represented by the isomerization coordinate RC. Rods have a remarkable property: They are so sensitive that they can reliably detect even the absorption of a single photon in darkness 4. The basis of this relationship, known as the Barlow correlation, has long been debated. The transition state mediating thermal activation has the same electronic structure as the photoreceptor excited state, thus creating a direct link between lambda(max) and k. 1225-1228 ISSN: 0036-8075 Subject: heat, isomerization, light, quantum mechanics, rhodopsin, rods (retina) Abstract: Responding to Light and Heat The protein rhodopsin is sensitive to dim light, but its sensitivity is limited by signals caused by the noise of thermal activation. Here we show that a quantum chemical model of the bovine rod pigment provides a molecular-level understanding of the Barlow correlation. The model suggests that when large amounts of R molecules are converted to.
#The molecular mechanism of dark noise in rod photoreceptors pdf#
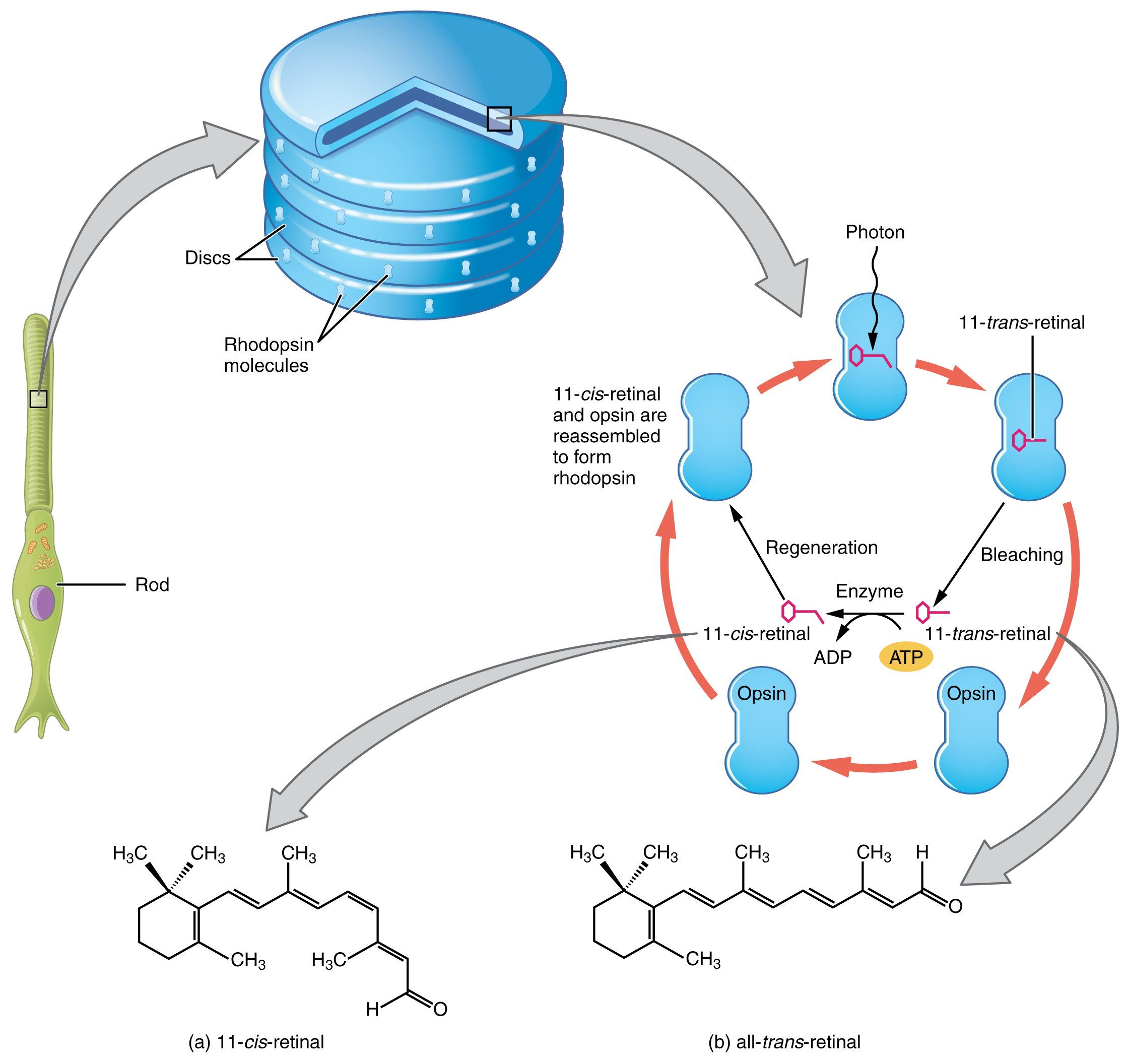
Nature minimizes photoreceptor noise by selecting a binding site geometry which shifts the p K a of the Schiff base proton to > 16, a value significantly larger than the p K a of the chromophore in bacteriorhodopsin (p K a ≈ 13) or model protonated Schiff bases in solution (p K a ≈ 7).The Molecular Mechanism of Thermal Noise in Rod Photoreceptors Author: Samer Gozem, Igor Schapiro, Nicolas Ferré, Massimo Olivucci Source: Science 2012 v.337 no.6099 pp. A molecular mechanism is suggested to account for the prolonged dark excitation. PDF Pugh highlights recent work ruling out a role for ultraweak photon emission in spontaneous photon-like events in retinal rods. This model predicts that the rate of photoreceptor noise will scale in proportion to 10 −ξ, where ξ is the p K a of the Schiff base proton on the retinyl chromophore. Subsequent research, in part inspired by Hecht et al.s discovery, has directly measured rod SPRs, characterized the molecular mechanism responsible for their generation, and uncovered much about the specializations in the retina that enable the reliable transmission of SPRs in the teeth of intrinsic neuronal noise. The credit of videoscopic surgery goes to this surgeon who had revolutionized the concept by making this instrument. 1953 The rigid rod lens system was discovered by Professor Hopkins. We conclude that this two-step process is likely to be the principal source of noise in all vertebrate and invertebrate photoreceptors. He also stressed the importance of continuous intra-abdominal pressure monitoring during a laparoscopic procedure. This mechanism is examined in detail and compared to other literature models for photoreceptor noise. Rod opsin is normally targeted to and retained in the membranes of the photosensitive outer segment through a C-terminal targeting sequence and by the presence of a periciliary barrier mechanism. Recent molecular orbital studies and pH experiments on horseshoe crabs ( Limulus) suggest that the thermal isomerization of a relatively unstable form of rhodopsin, one in which the Schiff-base linkage between the chromophore and protein is unprotonated, is responsible for thermal noise. A recent study suggested that thermal activation involves a canonical isomerization reaction. The random spontaneous events are strongly temperature dependent and in both vertebrate and invertebrate photoreceptors require activation energies usually in the range of 23 to 28 kcal mol −1. The protein rhodopsin is sensitive to dim light, but its sensitivity is limited by signals caused by the noise of thermal activation. Retinal photoreceptors generate discrete electrical events in the dark indistinguishable from those evoked by light and the resulting dark signals limit visual sensitivity at low levels of illumination.

